A complete platform for
real-world mobility research
From participant onboarding to sensor configuration, data ingestion and study-wide reporting —
Enoda provides an end-to-end platform to implement the Mobilise-D method.
Enoda provides an end-to-end platform to implement the Mobilise-D method.
Built on Mobilise-D, the world’s largest clinical mobility study
€50m
Over 5 years of research & trials
300+
Scientists, clinicians & industry experts
100+
Papers in leading journals
~2500
Participants in clinical trials
One platform, three purpose-built workspaces
Our platform is organised around three key roles that define trial success. Each role gets the tools they need — without the ones they don’t.
ROLE
01
Clinical Investigator
- Mange participant data
- Fill out forms
- Configure the sensor
- Upload data
ROLE
02
Data Scientist
- View data for individuals
- View study level data
- Assess changes
- Perform quality checks
ROLE
03
Project Manager
- Track key trial metrics
- Real time views of progress
- View activity logs
- Generate reports
Clinical-grade infrastructure, out of the box
Security, compliance and interoperability are table stakes for real-world research. Enoda handles them so your team doesn’t have to.
SECURITY
Encryption and controlled access
Study data is encrypted in transit and at rest, protected by SSO and role-based access controls. Data is served only through the platform API, with no direct database or storage exposure to the public internet.
RBAC
SSO
API-Only Access
COMPLIANCE
Audit-ready operations
Enoda is hosted in ISO 27001 compliant data centres, with audited suppliers and support for GDPR operations. Data is versioned, digitally signed and backed by audit trails for every modifying action.
ISO 27001
GDPR
Audit Trails
DATA RESIDENCY
EU-hosted by default
Study data is located in the EU by default, with hosting in Frankfurt. The platform can also support alternative regions or co-location in customer data centres, with continuous backups and personalised SLA support.
EU Default
Frankfurt
SLA Support
INTEGRATION
APIs, exports and pipelines
API services are available for integration with customer systems. Data can be exported in raw or CSV formats, and customer-specific code can be hosted as part of analytical pipelines.
API Services
CSV Export
Custom Pipelines
Simple browser-based access
Enoda runs securely through the browser, so study teams can access the platform without installing specialist software. Users only need an internet connection and a supported browser.
Mobilise-D out of the box. Anything else, your way
A core workflow built around the Mobilise-D protocols ships ready to use — but the platform is designed to bend around your study, not the other way around.
Mobilise-D, ready on day one. Custom workflows, ready by week one.
The default Enoda workflow is the Mobilise-D protocol — participant onboarding, sensor configuration, free-living capture and DMO generation, all pre-wired. From there, every step is configurable: visit schedules, eligibility logic, custom forms, validation rules and bespoke data fields can all be built to fit your study, securely.
One platform for every signal in your study.
Sensor streams are just the start. Collect eClinical Outcome Assessment (eCOA) responses, demographic data and bespoke outcomes — falls, blood pressure, anything you need — all linked back to the same participant.
Sensor streams
eCOA
Demographics
Outcomes
24 outcomes across 5 domains
Our comprehensive suite of digital mobility outcomes were validated across diverse patient populations.
Note: WB = walking bout. b-b = bout to bout. dur = duration. var = variability. WS = Walking Speed. L = Length.
Amount
Walking Duration
WB Step Count
Pattern
No. of WBs
No. of WBs > 10s
No. of WBs > 30s
No. of WBs > 60s
WB duration
P90 WB duration
WB dur. b-b var.
Pace
WS in 10-30s WBs
WS in >30s WBs
P90 WS in WBs>10 s
P90 WS in WBs>30 s
Stride L in 10-30s WBs
Stride L in >30s WBs
Rhythm
Cadence in all WBs
Cadence WBs >30s
P90 Cadence WBs >30s
Stride dur. in all WBs
Stride dur. in WBs > 30s
Variability
WS var. in WBs > 30s
Stride L var. WBs > 30s
Cadence b-b var.
Stride dur. b-b var.
Features that define the platform
Our platform delivers multiple features to simply workflows, automate processes and let you easily view data and manage the project.
MANAGE PARTICIPANTS
Automatically Process Data
Automated processes to configure the sensor in one click and automatically upload data straight to a dedicated participant folder. Minimise scope for errors and improve data quality.
Individual View
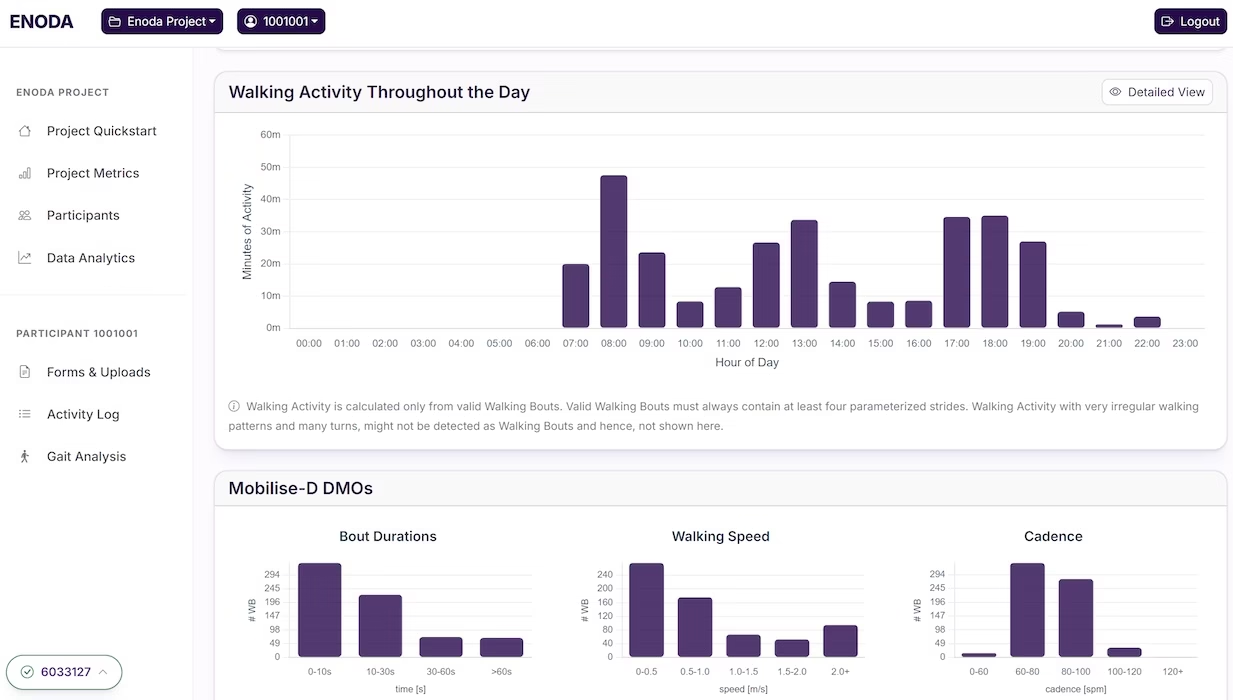
View activity throughout the day
We have a number of views that let you quickly and easily view walking activity throughout the day – whether that is in summary form or a visualisation of each individual walking bout.
Simple view
Detailed View
Walking Activity Throughout the Day
Assessment trend
m/s
Mean and P90 Walking Speed in Longer Bouts of >30 Seconds
Individual View
View changes in key metrics at multiple levels of granularity
Our platform lets you visualise changes in the DMOs over multiple levels of granularity – from assessment to assessment, from day to day and over the course of one day.
Study View
Results consolidated in our database
Individual results are automatically added to our database giving you a real-time view of the study as a whole. Visualise and download the study data whenever required.
Distribution
Metric
Walking Speed – WB >30s – Average
Stride duration
s
Stride duration (n = 1,459)
Study View
Data at whatever level of granularity you need
View, visualise and download data for individual walking bouts, as well as the aggregated data. Compare the distribution for any individual against the study as a whole.
Report View
Track key metrics
Track the progress of key metrics such as recruitment against your targets.
Recruitment
Target: 60
Recruitment Timeline
Submission progress
40% total
Form Submission Progress
Site
Northbridge Research Site
44 expected participants
Obtain participant consent
Timepoint 0 · 14 missing
30 / 44
68%
Notes associated with the T0 Visit
Timepoint 0 · 15 missing
29 / 44
66%
Pick a cohort
Timepoint 0 · 16 missing
28 / 44
64%
Axivity
Timepoint 1 · 23 missing
21 / 44
48%
Upload an Axivity Form
Timepoint 2 · 25 missing
19 / 44
43%
Demographic Information Form
Timepoint 1 · 30 missing
14 / 44
32%
MS Walking Scale Assessment
Timepoint 1 · 36 missing
8 / 44
18%
MS Walking Scale Assessment Form
Timepoint 2 · 41 missing
3 / 44
7%
Report View
View data submission progress
Track the progress of data collection and filter by key criteria such as the site, stage and the type of data. Export data as required for reporting purposes.
Validated and meaningful measures for clinical trials & clinical care
We work with both industry and academic researchers to deliver validated mobility data for your trial. We also work in the clinical care arena to deliver data that can assist in clinical decision making.
Planning a trial?
Talk to our team about how real-world mobility data can provide sensitive, meaningful and clinically validated data for your trial or clinical care programme
Want to learn more?
Read our whitepaper series on why real-world mobility matters and how to measure it.
